Farm productivity depends on nitrogen fertilisers.
Large-scale ammonia plants have dominated the industry and these use fossil fuels as a chemical feed stock and energy.
Other options are available - that use renewable feed stock and renewable energy to replace the fossil fuels used in traditional plants.
Their is also an opportunity to make smaller scale production plants that will allow fertiliser to be manufactured near to both the renewable feed stock and to the farms that use the fertiliser.
Urea is a widely used nitrogen fertiliser. Global production is estimated to have been 240 million tonnes in 2019.
Most is made with natural gas as a feed stock and a source of energy.
One tonne of urea has for much of the last ten to twenty years has cost about $500. It has risen sharply in price in 2021, costing around $1,500 a tonne.
One tonne of urea contains 200 kilograms of carbon.
Wheat straw and other crop waste containing cellulose contains this same amount of carbon in each 450 kilograms of cellulose.
The first step in making urea is to produce synthesis gas - a mixture of hydrogen and carbon monoxide.
This can be carried out with a device to gasify biomass.
An example is the gasifier made by All Power Labs which it builds to make synthesis gas it uses to power an engine to drive an electricity generator.
The PP30 Power Pallet is the culmination of our long work to create an expertly engineered, small-scale gasification solution that is realistic for today’s user. While personal scale gasification has long held tremendous promise, the realities of making it work usually prove too much for regular mortals. The high bar of operator expertise and extreme sensitivity to fuel particulars usually combine to make what seems simple in principle, exceedingly difficult in practice.
The Power Pallet has significantly widened this window for success by embedding the needed operator expertise in an onboard electronic brain.To increase the proportion of hydrogen in the synthesis gas - for a following ammonia production step - an electrolyzer that produces some hydrogen by electrolysis of water using renewable electricity - could also supply pure oxygen to use in the gasifier.
To convert the carbon monoxide to carbon dioxide and more hydrogen - both of which are used in later steps-
- hydrogen to react with nitrogen to produce ammonia, and
- carbon dioxide to react with that ammonia to produce urea
- a small-scale methane reforming unit is produced by Tokyo Gas that it uses as a fuel processing module in domestic fuel cell appliances.
 |
| Ene Farm Fuel Processing System by Tokyo Gas |
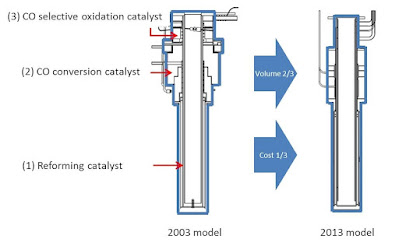
Because the three reactions take place at different temperatures, conventional practice is to use three varieties of reaction vessel. However, Tokyo Gas developed an integrated fuel processor that can handle the three chemical reactions in one vessel in 2000.
We set a mass production target for 2003, and subsequently achieved further structural streamlining, developed and improved a high-performance catalyst, and reviewed the catalyst operating method.
As a result, we succeeded in reducing total volume of the fuel processor by one third and production costs by two thirds in 2013.
The only other ingredient is nitrogen. Membrane filters are available that filter nitrogen from air for medium scale production processes.
A typical manufacturer of nitrogen membrane filters is Generon.
Since the first large-scale ammonia production plants were built, many technical enhancements have been identified in the equipment to maintain the optimum temperature and pressure, the design of catalysts, and the development of chromium-molybdenum steel reaction tubes. Each of these enhancements can be replicated in a scaled-down plant.
Small scale ammonia production plants are in operation that operate with renewable energy.
 |
| The Siemens green ammonia test plant uses wind power to convert hydrogen and nitrogen to ammonia. |
Siemens in the UK is working with researchers at the University of Oxford, the UK’s Science and Technology Facilities Council, and Cardiff University to run a demonstration plant using the typical Haber-Bosch process, powering it with wind. Ian Wilkinson, program manager in corporate technology at Siemens, names two reasons the firm chose to use only mature technology available today to run its plant.
First, Siemens wants to show that it can produce ammonia renewably, in a way that it can quickly scale up. The company also views the plant as a test system for ongoing technology development, including Haber-Bosch catalyst development and ammonia combustion tests.
The plan has worked so far. The small plant, set up in shipping containers, takes electricity from a wind turbine, runs it through a hydrogen electrolysis unit, and then uses the resulting hydrogen to synthesize ammonia.

0 comments:
Post a Comment